Gynecare Gynemesh Lawsuit: Suffer Mesh Complications From Ethicon Gynecare Gynemesh?
The Gynecare Gynemesh lawsuit represents a critical juncture in medical device litigation, underscoring the potential hazards associated with surgical mesh implants. As victims come forward with accounts of debilitating complications, including chronic pain and organ damage, the legal battles against Ethicon, the manufacturer, highlight a pressing need for accountability and enhanced patient safety measures. This discourse aims to explore the legal, medical, and ethical dimensions surrounding these cases. By examining the experiences of those affected and the responses from the legal system, we uncover the broader implications for medical device regulation and patient care. This examination invites a deeper consideration of the balance between innovation and patient welfare.
Mesh Complications Overview
Mesh complications, often arising from the use of products like Gynecare Gynemesh, have frequently led to severe health issues in patients, prompting a wave of lawsuits and legal actions against manufacturers. These lawsuits have highlighted the significant and sometimes debilitating complications associated with surgical mesh, including severe pain, infection, bleeding, organ perforation, and urinary problems. Many patients have undergone additional surgeries to remove or repair the mesh, yet some have suffered long-term consequences. The legal claims focus on allegations that the manufacturers failed to adequately warn patients and medical professionals about the risks and that the product was defectively designed. As a result, affected individuals have sought compensation for their injuries, medical expenses, and diminished quality of life, spotlighting a critical issue in medical device safety and accountability.
Gynecare Gynemesh Background

Introduced by Ethicon in 2002, Gynecare Gynemesh is a surgical mesh made from a non-bioabsorbable synthetic polymer designed for pelvic organ prolapse repair. This medical device aimed to provide a long-term solution for women suffering from pelvic organ prolapse, a condition where pelvic organs drop and press against the vaginal wall due to weakening of the pelvic muscles, often resulting from childbirth, surgery, or aging. The introduction of Gynecare Gynemesh represented a significant advancement in gynecological surgery, offering an innovative approach to reinforce the weakened vaginal wall. Ethicon, a subsidiary of Johnson & Johnson, marketed this product as a durable and effective solution for prolapse repair, emphasizing its potential to improve the quality of life for affected women.
Symptoms and Injuries Reported

Women who received Gynecare Gynemesh have reported a range of severe symptoms and injuries, necessitating medical attention and legal action. These complications include chronic pain, mesh erosion into the surrounding tissues, infections, and urinary problems. Some women have experienced a significant impact on their quality of life, including difficulties in sexual function and mobility issues. The non-bioabsorbable synthetic polymer material of the Gynecare Gynemesh, introduced in 2002, was intended for pelvic organ prolapse and stress urinary incontinence but has led to adverse outcomes. These reported symptoms and injuries have led to multiple surgeries for some patients, including mesh revision and repairs to damaged pelvic organs and tissues, highlighting the severe implications of mesh complications.
Legal Actions Against Ethicon

Given the severity of the injuries reported by patients using Gynecare Gynemesh, numerous legal actions have been initiated against Ethicon, the manufacturer. These legal actions spotlight the alleged failures of Ethicon to adequately warn patients and healthcare providers about the potential risks of using their product. These lawsuits, filed across various jurisdictions, contend that Ethicon's Gynemesh has caused significant and sometimes irreversible harm to patients. Plaintiffs seek justice for their suffering, which includes a range of complications from mesh erosion to chronic pain and infections. The legal battles against Ethicon aim to hold the company accountable for what claimants argue was the negligent design, manufacturing, and marketing of Gynemesh. This legal scrutiny underscores a critical examination of patient safety and corporate responsibility within the medical device industry.
Compensation Claims Process

Navigating the compensation claims process for injuries related to Gynecare Gynemesh involves several critical steps, including the gathering of medical evidence and legal representation. Victims must first secure comprehensive medical documentation that clearly links their injuries to the Gynemesh product. This often entails detailed medical records, diagnostic tests, and expert opinions. Following this, finding a lawyer experienced in medical device litigation, particularly with Gynecare Gynemesh cases, is essential. Such legal professionals are adept at navigating the complexities of these lawsuits, including negotiating settlements and representing clients in court if necessary. They also guide claimants through the stringent deadlines and procedural nuances specific to medical injury claims, ensuring that all necessary documentation is filed accurately and timely to maximize the chances of a favorable outcome.
Eligibility for Lawsuit Participation

Determining whether an individual is eligible for lawsuit participation requires a thorough evaluation of their medical history and the specific circumstances surrounding their use of Gynecare Gynemesh. Criteria for eligibility typically encompass having undergone implantation of the Gynemesh product, experiencing complications or adverse effects directly attributed to the mesh, and having evidence of such issues through medical records or expert testimony. Additionally, the timeframe within which the mesh was implanted and the complications arose plays a crucial role in establishing a connection to the Gynecare Gynemesh. Legal statutes of limitations also significantly impact eligibility, as they dictate the period within which a claim must be filed. Consulting with a legal professional skilled in medical device litigation is imperative for a comprehensive assessment of eligibility.
Impact on Quality of Life

Many women who have undergone implantation with Gynecare Gynemesh have reported significant adverse impacts on their quality of life due to complications arising from the mesh. These complications can range from chronic pain, which limits daily activities and workability, to emotional and psychological distress stemming from ongoing health concerns and bodily changes. In some cases, the severity of the mesh complications has led to a decrease in social interactions and strained personal relationships, further diminishing the overall well-being of the affected individuals. The physical discomfort and the uncertainty regarding future health outcomes have also been reported to contribute to anxiety and depression, marking a profound and multifaceted impact on the quality of life of those affected by Gynecare Gynemesh complications.
Understanding Mesh Revision Surgeries
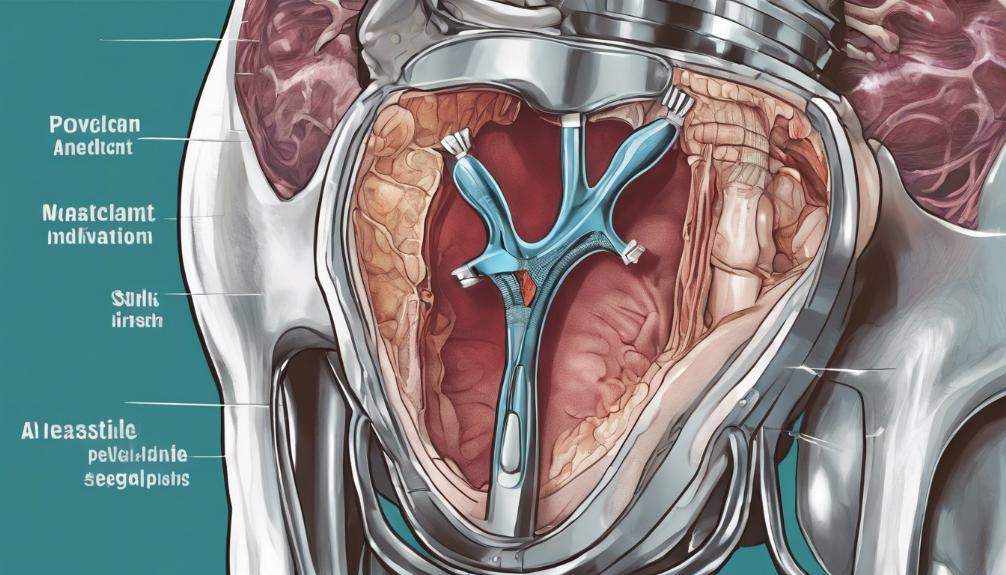
Mesh revision surgeries are crucial interventions aimed at addressing the complications that arise from the implantation of devices like Gynecare Gynemesh. These surgeries are often necessitated by issues such as mesh erosion, infection, chronic pain, or organ perforation. The procedure involves the removal or correction of the problematic mesh to alleviate symptoms and prevent further damage to pelvic organs and tissues. Due to the complexity of such surgeries, they require skilled surgeons experienced in pelvic floor reconstruction. The goal is to restore the patient's quality of life while minimizing the risks of recurrence of the complications. For many women, undergoing mesh revision surgery is a critical step towards recovery from the adverse effects of mesh implantation.
Other Mesh-Related Lawsuits

In addition to the Gynecare Gynemesh cases, there are numerous lawsuits related to other mesh products that have led to significant health complications for patients. These lawsuits span a variety of applications, including transvaginal mesh procedures for pelvic organ prolapse and stress urinary incontinence, as well as hernia repair operations. Many of these cases center around allegations of product design flaws, inadequate warning of potential risks, and failure to test the products properly before market release. The complications reported range from chronic pain, infection, erosion of the mesh into surrounding tissues, to the necessity for additional surgeries to remove or repair the mesh. These legal actions have brought attention to the safety and regulatory approval processes of medical devices, prompting calls for more rigorous testing and transparency from manufacturers.
Seeking Legal Assistance

Individuals experiencing complications from medical devices or medications may benefit from consulting with legal professionals to explore potential compensation avenues. Legal counsel specializing in medical injury cases can provide valuable insights into the feasibility of a lawsuit based on the specifics of an individual's situation. They can navigate the complexities of legal claims associated with products like Gynecare Gynemesh, ensuring that affected parties understand their rights and the steps necessary for pursuing justice. Moreover, legal experts can assist in gathering evidence, filing claims within statutory deadlines, and representing individuals in court if necessary. Seeking professional legal assistance can be a crucial step for individuals seeking redress for their injuries and holding responsible parties accountable for their actions.
Settlements and Verdicts

Exploring the legal landscape further, settlements and verdicts in cases involving medical devices and drug injuries offer a glimpse into the judicial outcomes for affected individuals. In the realm of Gynecare Gynemesh lawsuits, these outcomes often reflect the severity of injuries sustained and the impact on patients' lives. Successful claims have led to compensation for medical expenses, pain and suffering, and lost wages. While specific settlement amounts and verdicts vary widely, they underscore the legal system's role in addressing harm caused by medical products. These outcomes not only provide financial relief to victims but also signal to manufacturers the importance of safety and accountability in product development and marketing, setting a precedent for future litigation in similar cases.
Preventing Future Complications

To mitigate the risk of mesh-related complications in the future, it is essential for healthcare providers to rigorously assess the safety and efficacy of medical devices before recommending their use to patients. This entails a thorough review of clinical data, potential side effects, and long-term outcomes associated with such devices. Furthermore, personalized patient assessments should be conducted to ensure that the benefits of using a particular medical device outweigh the risks for that individual. Continuous education and training on the latest advancements and safety protocols in the field of medical devices are crucial for healthcare professionals. Additionally, patients should be adequately informed about the possible risks and encouraged to report any adverse symptoms promptly, enabling timely intervention and reducing the likelihood of severe complications.
Resources for Affected Individuals

Several resources are available to assist those impacted by mesh complications, offering guidance on legal recourse and support services. Affected individuals can access specialized legal firms experienced in handling Gynecare Gynemesh and other related lawsuits. These firms often provide free consultations to evaluate the merits of a case and advise on the potential for compensation. Additionally, support groups and online communities offer emotional and practical support, enabling those affected to share experiences and coping strategies. Medical professionals specializing in mesh complications can also provide essential health advice and treatment options. Furthermore, informational websites and legal advocacy groups offer updates on litigation, research, and policy changes related to mesh products, ensuring affected individuals remain informed and empowered to pursue justice and recovery.
Frequently Asked Questions
How Has the Public Perception of Mesh Implants Changed Since the Initiation of Gynecare Gynemesh Lawsuits?
Since the initiation of lawsuits related to mesh implants, public perception has significantly shifted. Previously seen as a relatively safe and effective solution for various medical conditions, these implants are now approached with caution. The increasing awareness of potential severe complications and the subsequent legal actions have highlighted the risks associated with their use. This change in perception is reflected in both patient decision-making and the broader medical community's recommendations regarding mesh implant treatments.
What Are the Psychological Effects on Patients Dealing With Long-Term Complications From Gynecare Gynemesh Implants?
Patients experiencing long-term complications from mesh implants often face significant psychological effects. These can include chronic pain, anxiety, depression, and a diminished quality of life. The psychological burden is compounded by the physical discomfort and the potential need for additional surgeries or treatments. It's crucial for healthcare providers to address not only the physical but also the mental health aspects of patients enduring such complications, offering comprehensive care and support.
Has There Been Any Significant Medical Advancements or Alternatives Introduced in Response to the Gynecare Gynemesh Controversy?
In response to complications associated with pelvic mesh devices, significant medical advancements and alternatives have been developed. These include less invasive surgical techniques, the introduction of biocompatible materials for mesh production, and enhanced pre-surgical assessment protocols to better identify suitable candidates for mesh implantation. These advancements aim to reduce the occurrence of complications and provide safer, more effective treatment options for conditions such as pelvic organ prolapse and stress urinary incontinence.
How Do Gynecare Gynemesh Lawsuits Compare to Other Medical Device Lawsuits in Terms of Complexity and Duration?
Gynecare Gynemesh lawsuits, in comparison to other medical device lawsuits, often exhibit a similar level of complexity and can span extensive durations. These cases involve intricate medical evidence and substantial legal arguments regarding product safety, efficacy, and manufacturer liability. The need for expert testimonies, detailed patient medical histories, and the assessment of long-term health impacts contribute to the complexity and length of litigation, paralleling challenges seen in other medical device legal battles.
What Role Do Patient Advocacy Groups Play in Supporting Individuals Affected by Gynecare Gynemesh Complications?
Patient advocacy groups play a crucial role in supporting individuals who have experienced complications from medical devices, including mesh products. These organizations provide vital resources, including information on legal rights and access to specialized medical care. They also foster a community for shared experiences and support, elevate awareness of potential complications, and advocate for stronger regulatory measures. Their involvement is instrumental in guiding affected individuals through recovery and legal processes for compensation.

This post has been generated by AI and was not reviewed by editors. This is Not legal advice. Please consult with an attorney.




